Accelerate medical device development with our end-to-end expertise
Trusted by industry leaders
Trusted by industry leaders

For enterprise teams and high-growth startups. Confidential. No obligation.
In highly regulated industries like MedTech, Delve helps accelerate innovation and avoid costly missteps. We offer:
Our integrated team of expert strategists, designers, engineers, and human factors practitioners will validate real-world feasibility and usability through early user research, concept iteration, and testing.
See how we helped create a user experience that’s intuitive and painless for Dexcom.
Delve will guide your team through every project milestone, including regulatory and compliance stage gates, to increase your odds of success and help you bring your product to market faster with no requirement missed.
Learn how we supported a successful launch for Otoscan.
We embed regulatory, design for manufacturability, and clinical user requirements into every phase of the product development process.
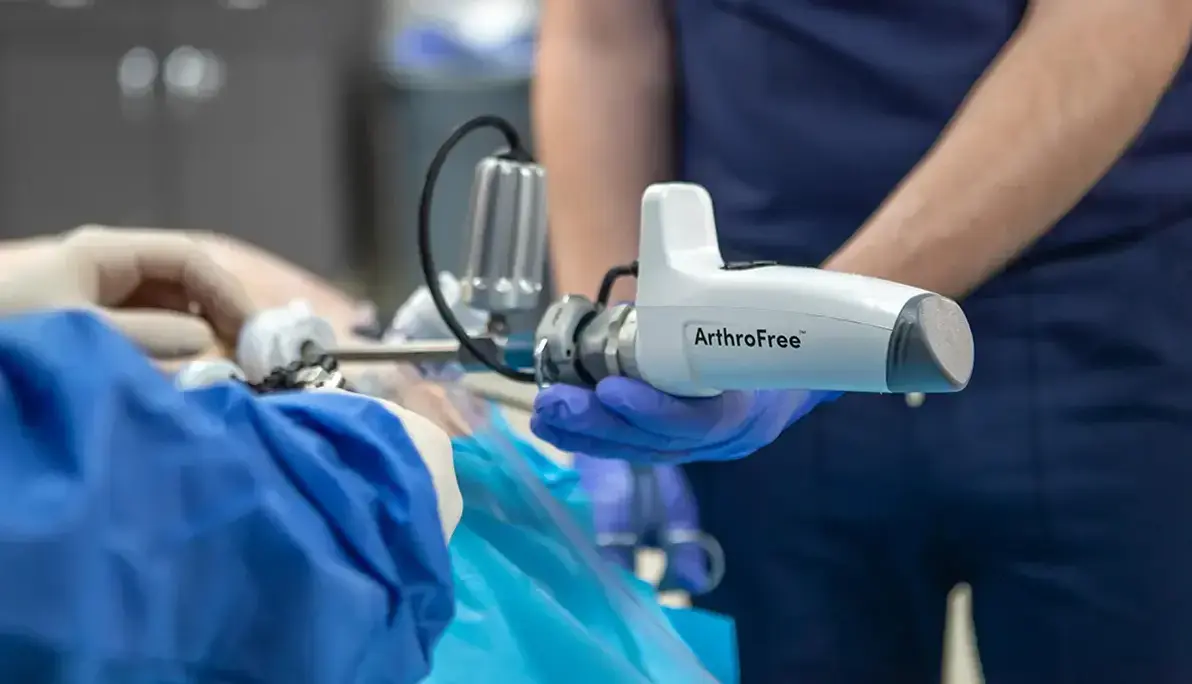
See how we helped to bring Lazurite’s ArthroFree to market.
Delve ensures cross-functional alignment from project kickoff to launch with a proven orchestration process for high-stakes medical device development programs.
Take a closer look at how we redesigned the overall experience of the ProNova SC360.
.webp?width=776&height=407&name=06_Work_Case_Study_Dexcom_G6_Applicator_G5Comparison%20(1).webp)
Dexcom G6 Continuous Glucose Monitoring device - See case study



Delve’s expertise in user research and design significantly improved our product’s user experience. Delve brings strengths that we don’t have, which is why I’m looking forward to working with them again.
They were really passionate about making something that looked beautiful and behaved in beautiful ways.
The technical innovation in Otoscan combined with unprecedented simplicity makes customization a great experience for our patients and audiologists.
Working with our UX lead, the design team came up with a wonderful visual interface with intuitive designs for certain features. This has helped triple engagement in the platform.

Delve helped us define a patient environment that is a true differentiator for our new company.

How Dexcom turned a complex clinical task into a simple, everyday action—and unlocked mass adoption in the process.
In order to better serve the needs of people with Type 2 diabetes, BD set out to create a central, digital hub for people to manage all aspects of their care.
A device that is used in the neonatal critical care environment that is designed to reduce device-related workflow.
Delve helped BK Ultrasound bundle its game-changing technology into a widely adopted, easy to use, award-winning medical device.
Define opportunities grounded in user needs, technical feasibility, and market context. Foundational research and framing to help define your opportunities for innovation.
Explore and evaluate concepts with speed, supported by CNC, 3D printing, and integrated design-engineering teams at all locations.
From formative studies to summative usability testing, we help you meet HFE/UE expectations for Class I, II, and III devices.
Optimize product development for scale, reliable production, and supply chain stability.
We align software with hardware design, compliance standards, and user experience across embedded, mobile, and cloud systems.
Delve brings ISO 13485, IEC 62366, and FDA compliance expertise with deep experience in documentation and quality control for medical devices.
